60. Selenide-Enabled Photocatalytic Hydroazolation of gem-Difluoroalkenes
Abd El Gaber M. K.; Herrick, R.; Sudhakar, P.; Rana, A.; Roach, B. A.; Dick, J. E.; Altman, R. A.* Chem. Sci., 2025.
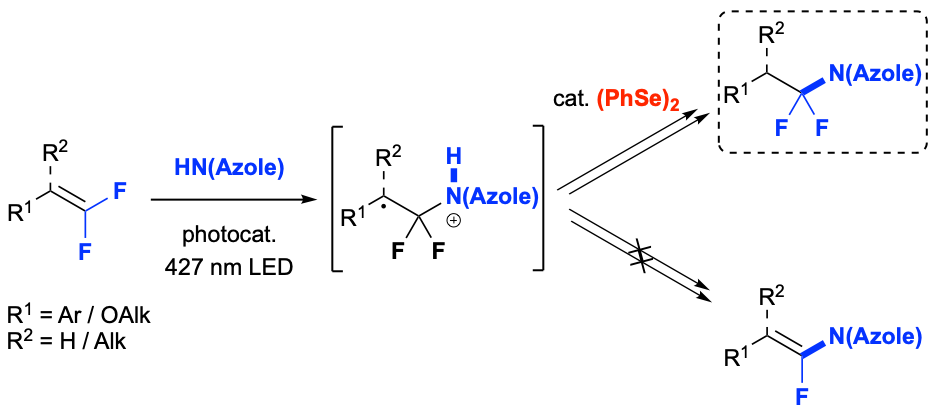
Difluoromethylene-containing molecules and azoles, independently, have wide applications in materials science, pharmaceuticals, agrochemicals and as biological diagnostic probes. However, compounds bearing the N-α,α-difluoroalkyl azole [(azole)N–CF2R] motif remain scarce in academic and patent literature, presumably due to a lack of synthetic methods. Such compounds could be convergently accessed in a single step via the hydroazolation of gem-difluoroalkenes. However, most existing functionalization reactions of gem-difluoroalkenes proceed through a β-fluoride elimination pathway that generates monofluorinated derivatives. Herein, we report a photocatalytic hydroazolation of gem-difluoroalkenes to generate (azole)N–CF2R that employs an uncommon diselenide co-catalyst to avoid the defluorinative process, ultimately enabling facile access to underexplored medicinally and agriculturally-relevant chemical space.