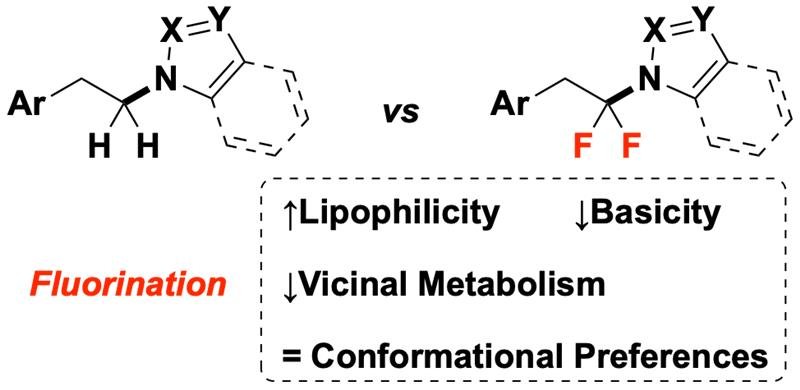
62. The Effect of N-Alkyl Azole Difluorination on Molecular Properties Relevant for Lead Optimization: A Comparative Study
Herrick, R. M.; Green, S. A.; Rich, S.; Grandner, J. N.; Huard, K.;* Altman, R. A.* “The Effect of N-Alkyl Azole Difluorination on Molecular Properties Relevant for Lead Optimization: A Comparative Study” ACS Med. Chem. Lett.2026 10.1021/acsmedchemlett.5c00757.
Despite recent interest in N-trifluoromethyl azoles, N-α,α-difluoroalkyl azoles [(azole)N–CF2R] remain understudied and underutilized in medicinal chemistry. To address this deficiency, we have conducted a comparative study of medicinally relevant properties for a series of (azole)N–CF2R and their nonfluorinated matched molecular pairs (MMPs) that revealed fluorine-induced reductions in azole pKa, hydrophilicity, experimental polar surface area, and metabolic oxidation of a labile vicinal position. Additionally, computational analysis supports the fluorine-induced suppression of metabolic aliphatic oxidation but suggests a limited impact of fluorination on conformational preferences within MMPs. Along with a newly provided synthetic method to install such a substructure, this information will facilitate rational incorporation of (azole)N–CF2R groups in drug optimization campaigns.